Biomaterials may form, evolve and/or degrade differently when its composition, formulation or preparation changes. This platform will help you to evaluate the unique behavior of your biomaterial.
Non-destructive viscoelasticity measurement of biomaterials
The ElastoSens Bio is a non-destructive contactless technology to assess the viscoelastic properties of different materials. It is a cell-friendly and easy-to-operate system for analytical testing of soft biomaterials providing consistent and repeatable results. Additional features like the Peltier temperature control, the photostimulation source or the built in gas entries for environmental control allows cell testing over a longer period of time to measure not only the degradation of hydrogels in natural tissues and drug release but also the stiffness of extracellular matrices.
Learn more and download our application note "Measuring the efficacy of hemostatic agents using ElastoSens Bio"

Measure kinetics of blood coagulation and hemorheology
Conventional techniques cannot possible to measure absolute blood viscoelastic properties during coagulation. The non-destructive measurement principle of the ElastoSens Bio has shown to provide repeatable and sensitive measurements to test blood coagulation through the viscoelastic properties of the forming clot. The concentration of the CaCl2 or other hemostatic agents (HA) in blood affects the initialization of coagulation (showing a non-linear behavior). The stiffness of the developing clot is proportional to the concentration of the HA. This technology provides a quantitative method for better investigation of the kinetic of blood coagulation. Conventional and more recent techniquesl like thromboelastography (TEG), rotational thromboelastometry (ROTEM) and magnetomotive optical coherence elastography do not directly measure the absolute viscoelasticity of the clot.
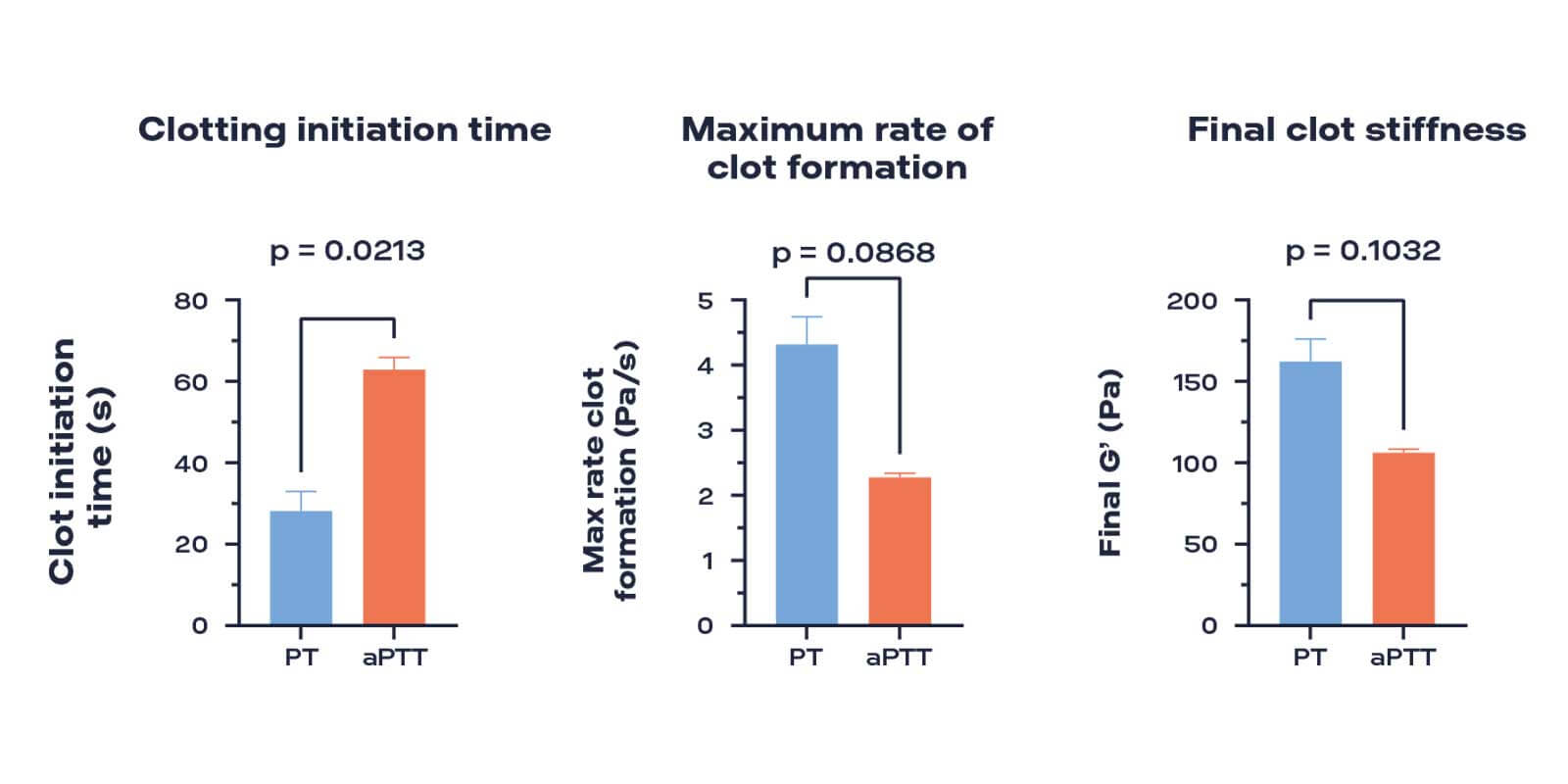
Clotting initiation time, maximum rate of clot formation, and final clot stiffness of calf plasma with PT (blue) and aPTT (orange) reagents.
Learn more and download our application note "Viscoelasticity characterization of coagulating plasma using ElastoSens Bio"
Hydrogel formulation
Cellularized hydrogels have been widely investigated for producing in vitro models of tissues such as skin, blood vessels, bone, etc. These models have a high potential for an alternative to animal models used in trials for studying physio-pathological processes and for testing new drugs and medical devices.
The formulation of hydrogels, including parameters such as polymer concentration and crosslinking agent, needs to be optimized for each specific application, dependent on the source of cell type. Conventional techniques used for assessing the mechanical properties of hydrogels such as rheometer, tensile and compression tests are often destructive and prevent the re-use of samples for further characterizations. Furthermore, long term studies over days or weeks require multiple samples which can be a major limiting factor specially when they contain cells. The ElastoSens Bio allows to measure viscoelastic properties during gel formation in a non-destructive way by keeping the samples between tests in a biological incubator. This allows an easy application of various conditions to the biomaterial in order to simulate in vivo environments. For example, the evaluation of the mechanical stability during 14 days of gelatin hydrogels seeded with keratinocytes with the ElastoSens™ Bio assisted in the selection of the gel formulation used in the epidermis model.
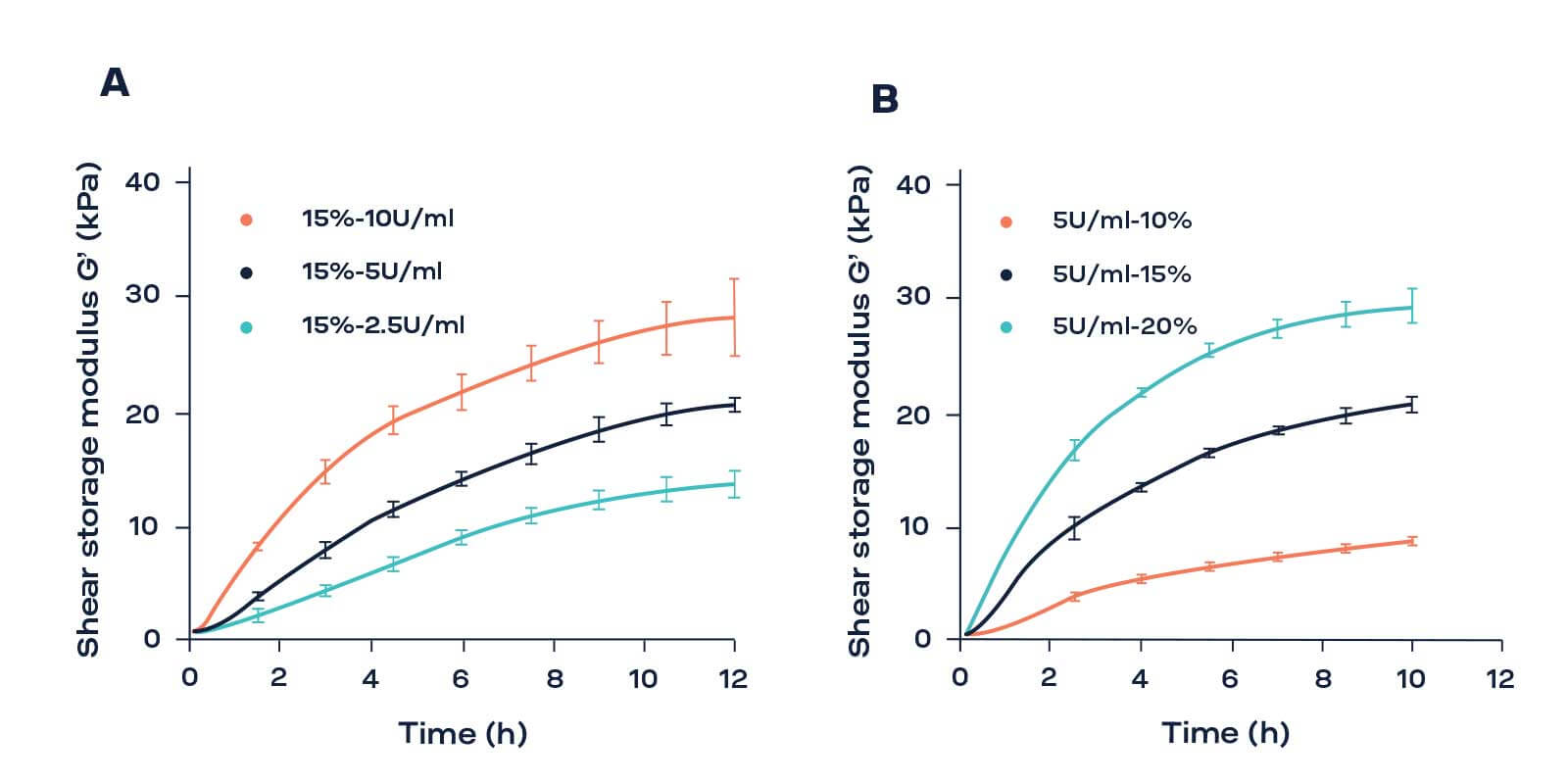
A) The evolution of the shear storage modulus (kPa) as a function of time of 15% (w/w %) gelatin crosslinked with 2, 5, and 10 U/mL of TG.
B) The time evolution of the shear storage modulus (kPa) of 10 %, 15 %, and 20 % (w/w %) gelatin with 5 U/mL of TG.
Learn more and download our application note "Gel formulation and long term mechanical stability analysis of cellularized gelatin-based hydrogel using ElastoSens Bio"
Degradation and Drug release of hydrogel based drug delivery systems
The controlled release of drugs at precise locations within the body can prevent systemic toxicity and deliver accurate dosages to patients. One of the release mechanisms of therapeutic agents is based on the controlled degradation of the hydrogel that carries the drug. In this case, the decrease in the viscoelastic properties of the hydrogel is proportional to the amount of released drug. Conventional testing instruments are difficult to use difficult to use for evaluating the mechanical properties of hydrogel-based drug release systems during degradation. Because destructive technique, they prevent the re-testing of the same hydrogel sample for long term degradation studies. This leads to the need of more samples to monitor a complete drug release study. Compared to these limitations of conventional methods, the ElastoSens™ Bio is able to capture subtle mechanical changes during degradation before the sample starts to disintegrate. It provides precise measurements of the mechanical properties of dye or drug loaded hydrogels, dependent on the temperature. The ElastoSens Bio can be used to develop or formulate hydrogels to analyze their degradation profile and to test and optimize degradation mechanisms, by enzymatic or chemical reactions. The resulting viscoelastic measurements allows a correlation to the amount of drug release.
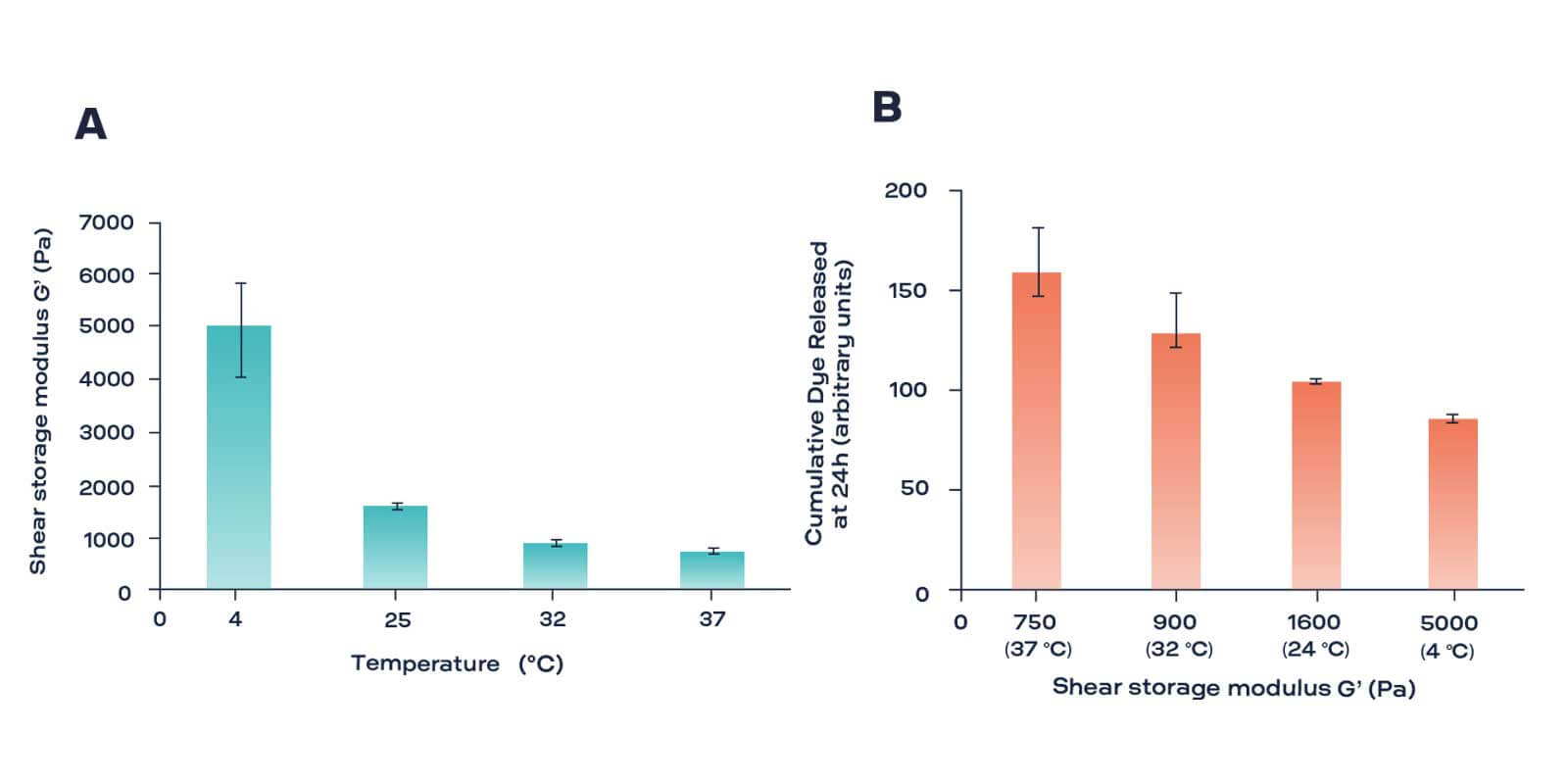
Storage modulus (G’) of alginate/gelatin gels at different temperatures (left) and total dye released from composite gels at 24 hours relative to average G’ (right).
Learn more and download our application note "Degradation and drug release of hydrogel-based drug delivery systems"
Measure non-destructive the viscoelasticity of biomaterials with the ElastoSens Bio!
ElastoSens™ Bio is a viscoelasticity testing platform that uses vibrations to obtain non-destructive contactless viscoelastic measurements of soft matter.
- Benchtop device with small footprint
- Non-destructive contactless viscoelasticity testing of soft materials
- Real time measurement of swelling and degradation of biomaterials
- Control of up to 5 instruments by a single tablet App via wireless communication
- Photostimulation option of 3 built-in UV sources for customized measurement sequences
- Advanced temperature control option for setting up customized temperature profiles for testing


